2025-05-09
Gene editing technology, as a revolutionary breakthrough in modern biomedicine, is profoundly reshaping our understanding of life sciences. Since the introduction of the CRISPR-Cas9 gene editing tool in 2012, gene editing has rapidly become a research hotspot in the life sciences. By mimicking bacterial immune mechanisms, this technology uses specific RNAs to guide the Cas9 protein to cut at precise DNA locations, thereby enabling accurate genetic modifications. In recent years, continuous developments of gene editing technology have further expanded its applications.
The delivery vectors for gene editing tools are a critical component in ensuring the success of gene editing, as they are responsible for transporting editing components (such as Cas9 and gRNA) into target cells. The choice and application of delivery vectors directly affect editing efficiency and safety. Currently, the most representative vectors for gene editing delivery include viral vectors (e.g., adeno-associated virus, AAV) and non-viral vectors (e.g., lipid nanoparticles, LNPs). However, each comes with its own limitations. For instance, AAV vectors may lead to increased off-target risks and immunogenicity due to prolonged Cas9 expression, while LNPs, despite their favorable safety profile, primarily accumulate in the liver, limiting delivery efficiency to extrahepatic tissues.
Against this backdrop, virus-like particle (VLP) technology has emerged as a promising next-generation delivery platform due to its unique advantages. VLPs are particle structures self-assembled from viral capsid proteins that resemble viruses in appearance and function but lack viral genetic material. As such, they combine the benefits of both viral and non-viral vectors, making them highly attractive for delivering gene editing tools. In gene editing, VLPs used to deliver mRNA or proteins are mostly derived from lentiviruses or retroviruses. Compared with other delivery vectors, VLPs offer several advantages:
Loading flexibility: Immature retroviral particles lack rigid symmetric structures, allowing greater flexibility in accommodating payloads. With a diameter of 100–200 nm, lentivirus/retrovirus-based VLPs are compatible with various payload forms, including mRNA and RNPs.
Targeting plasticity: Tissue-specific delivery can be achieved through viral envelope glycoprotein modification.
Low off-target effects: Transient delivery significantly reduces the risk of off-target editing.
High safety: As VLPs are composed entirely of proteins and lack DNA, they avoid the risk of viral genetic material integrating into the host genome.
Genevoyager one-stop VLPs solutions
Genevoyager is committed to delivering one-stop VLP services for the academic community, pharmaceutical companies, and biotechnology companies worldwide. Genevoyager has established a comprehensive technology platform covering the entire process from basic research to preclinical applications, supporting engineered modification and customized production of VLPs. Services include, but are not limited to, VLP purification, targeted modification, multidimensional characterization, and fully customized CRO solutions.
The breakthrough of the team led by Yujia Cai: VLP-based mRNA delivery technology
In 2018, the research team led by Yujia Cai developed a lentiviral vector-based technology for delivering exogenous RNA (Patent No. CN108504689A). This technology exploits the principle of specific binding between the RNA-binding protein MS2 and its cognate RNA sequence (MS2 stem loop). The MS2 protein was incorporated into the structural backbone of the lentiviral GagPol polyprotein, while the MS2 stem loop sequence was fused to the exogenous target RNA. As a result, the target RNA could be packaged into lentiviral particles during viral assembly. In this approach, the MS2 protein was fused with the lentiviral Gag and positioned at the N-terminus, while the MS2 stem loop was placed at the N-terminus of EGFP, thereby achieving higher delivery efficiency, as illustrated in the figure below.
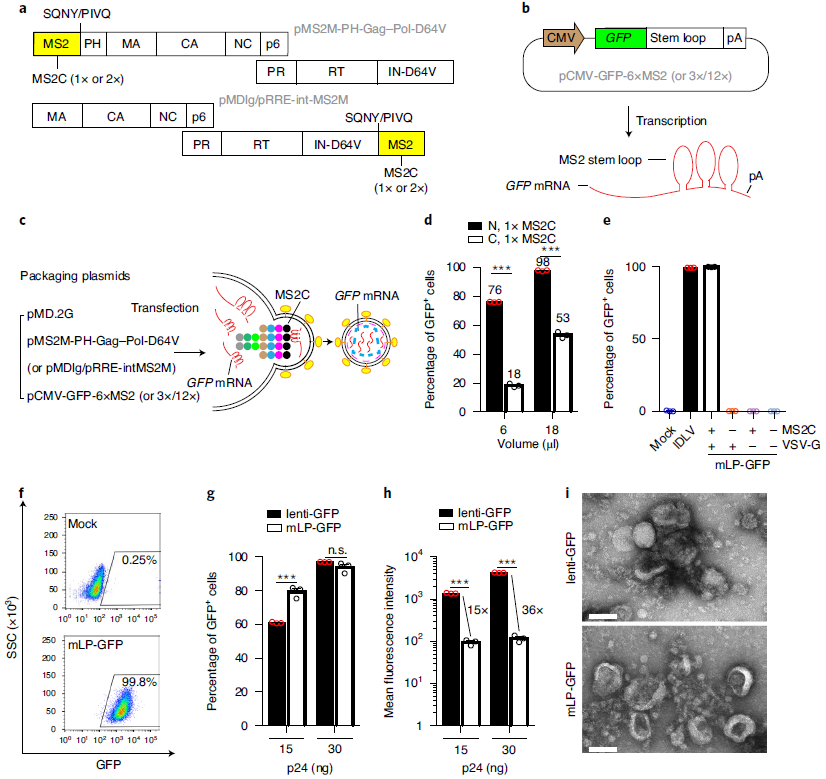
Figure 1: Construction of a lentiviral system for efficient mRNA delivery
(Source: Ling S et al, Nat Biomed Eng, 2021)
Based on this technology, the team led by Yujia Cai published research articles in Nature Biotechnology and Nature Biomedical Engineering in 2021 to use VLPs to deliver Cas9 mRNA and sgRNA to achieve the effective delivery of gene editing. The Cas9 delivered with this technology was expressed for only 3 days in vivo, which might minimize the off-target activity of the long-term Cas9 expression. This technology also showed good performance in herpetic stromal keratitis (HSK) treatment. Subsequently, the team led by Yujia Cai translated this technology into the development of BD111, which was granted Orphan Drug Designation (ODD) by the U.S. FDA in June 2022. To date, Phase I clinical administration has been completed in three patients with recurrent type I herpes simplex stromal keratitis. Preliminary clinical observation and evaluation over 18 months revealed no serious adverse events, and the patients responded well. The figure below illustrates the design of BD111 and its antiviral mechanism.
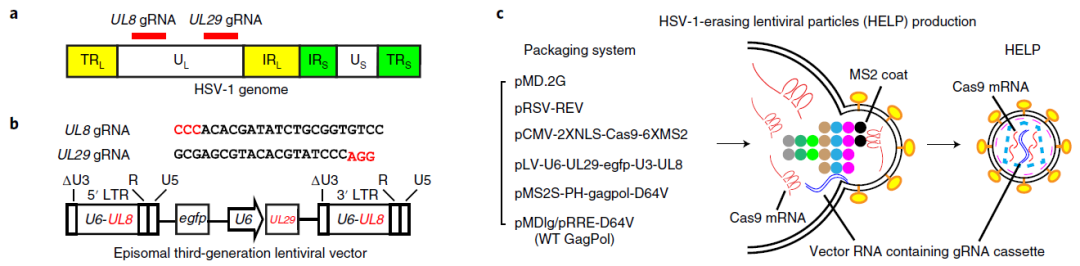
Figure 2: The design of BD111
(Source: Yin D et al, Nat Biotechnol., 2021)
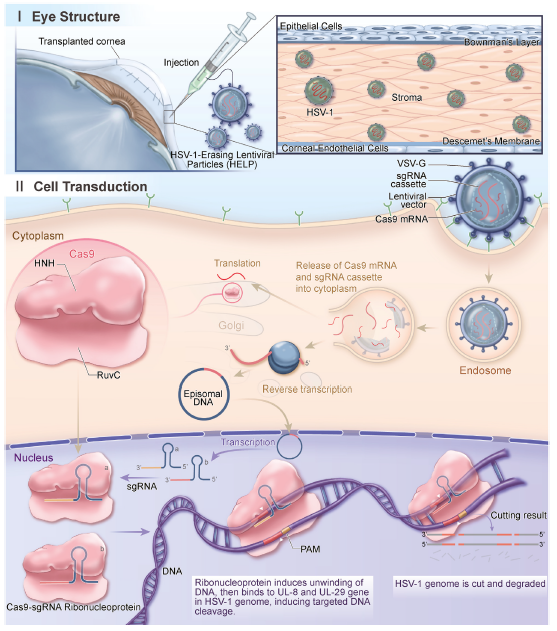
Figure 3: The antiviral mechanism of CRISPR in human corneas
(Source: Wei A et al, Mol Ther., 2023)
The new mRNA-delivering system developed by the team led by Feng Zhang: Selective Endogenous eNcapsidation for Cellular Delivery (SEND)
In 2021, the team led by Feng Zhang published a research article in Science about developing Selective Endogenous eNcapsidation for Cellular Delivery (SEND) to package, secrete, and deliver specific RNAs. SEND is based on the RNA-delivering protein PEG10 that naturally exists in humans. This protein is a gag homolog and the LTR retrotransposon-derived protein that binds and facilitates vesicular secretion of its own mRNA. To develop PEG10 into a delivery platform, the researchers reprogrammed this protein. First, they identified the sequence that recognizes and packages its RNA in the mRNA sequence of PEG10. Then they reprogrammed PEG10 and this mRNA sequence to selectively package RNA. To intensify the ability of the PEG10 system to enter cells, the researchers modified PEG10 with VSVg and SYNA, endowing it with the ability to infect a wide range of cell types. Since PEG10 is a naturally occurring human protein, this platform can effectively evade immune attacks compared with other RNA delivery approaches.
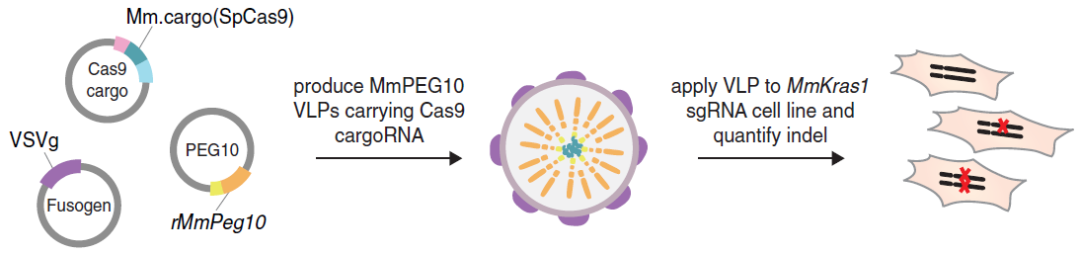
Figure 4: The retooling of SEND for genome engineering
(Source: Segel M et al, Science, 2021)
The team led by David Liu achieved a breakthrough in delivering base editors (BEs) and prime editors (PEs) using engineered virus-like particles (eVLPs)
In 2022, the team led by David Liu published a research article in Cell. They systematically optimized the factor that restricts the efficiency of delivering BE-VLPs and developed fourth-generation eVLPs. BE RNPs loading within v4 eVLPs was improved by an average of 16-fold compared with initial designs based on previously reported VLP scaffolds. The in-vivo experiment results show that the editing efficiencies of v4 BE-eVLPs were substantially improved in HEK293T cells to 95% at the maximal dose, in NIH 3T3 cells to 80%, and in primary human and mouse fibroblasts to 90%. Meanwhile, v4 eVLPs substantially reduce off-target editing. In addition, a 63% editing in the mouse liver and a 78% reduction in serum Pcsk9 protein levels were observed following treatment with v4 BE-eVLPs. Moreover, the BE delivered by eVLPs partially restores visual function in a mouse model of genetic blindness.
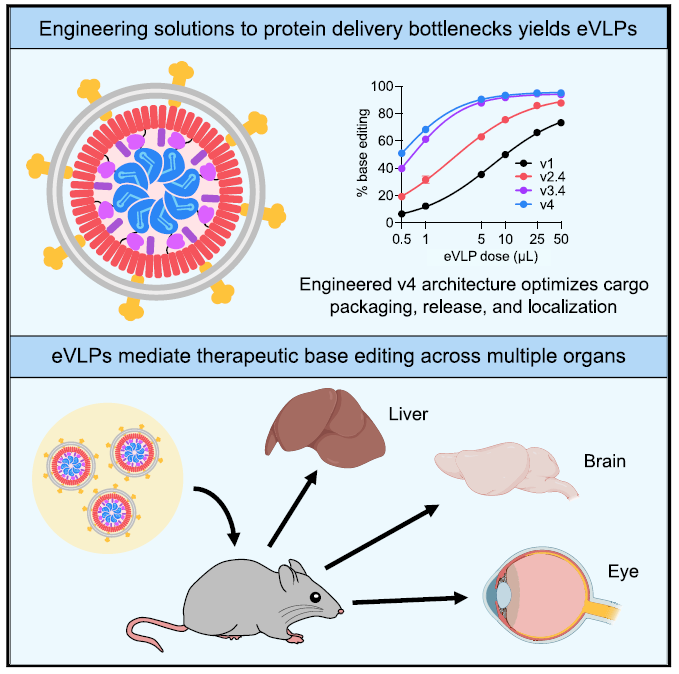
Figure 5: Engineered virus-like particles achieve base editing across multiple organs
(Source: Banskota S et al, Cell, 2022)
In their latest research, this team combined v4 eVLPs and prime editor (PE), an engineered protein consisting of a catalytically impaired programmable nickase domain (such as a Cas9 nickase) fused to an engineered reverse transcriptase (RT) domain, which can precisely achieve genome replacement, insertion and deletion in mammalian cells. They made further improvements to it and developed PE-eVLPs. Compared with v1 PE-eVLPs, third-generation v3 PE-eVLPs offer a 79-fold improvement in prime editing efficiency in mouse Neuro-2A (N2A) cells and a 170-fold improvement in human HEK293T cells. In vivo experiment results show that v3 PE-eVLPs could correct a 4-bp deletion in Mfrp in the rd6 mouse model of retinal degeneration (15% average efficiency) and correct an Rpe65 substitution to partially rescue visual function in the rd12 model for human Leber congenital amaurosis (LCA) (7.2% average efficiency).
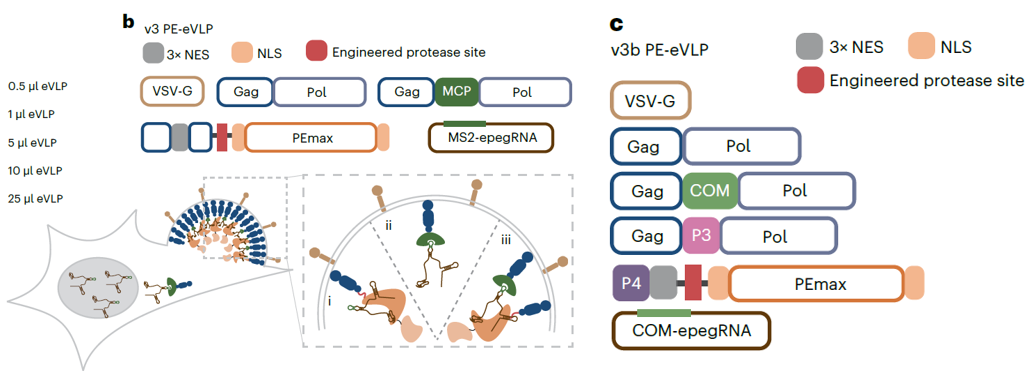
Figure 6: Schematic of v3 PE-eVLPs utilizing the MCP–MS2 strategy for the recruitment of epegRNAs and the v3b PE-eVLP system
(Source: An M et al, Nat Biotechnol.,2024)
The team led by Jennifer A. Doudna successfully simplified the VLPs delivering system: miniEDV
Although VLPs have shown broad application prospects in the field of gene therapy, they are derived from lentiviruses/retroviruses and retain a substantial amount of viral protein components, meaning their intrinsic safety still requires further optimization. In December 2024, the team led by Jennifer A. Doudna published a research article in PNAS. With systematic mechanism research, they successfully developed a minimal EDV (miniEDV) that substantially reduces production complexity while retaining full RNP delivery capability.Compared with traditional VLPs, MiniEDVs are 25% smaller and ~80% of viral residues were removed, yet it’s package equivalent amounts of Cas9 RNPs. In vivo experiment results show that in primary human T cells, MiniEDVs packaging increased editing by 53% to 107%. An average increase in editing per EDV particle of ~2.5-fold in HEK-293T is also observed.
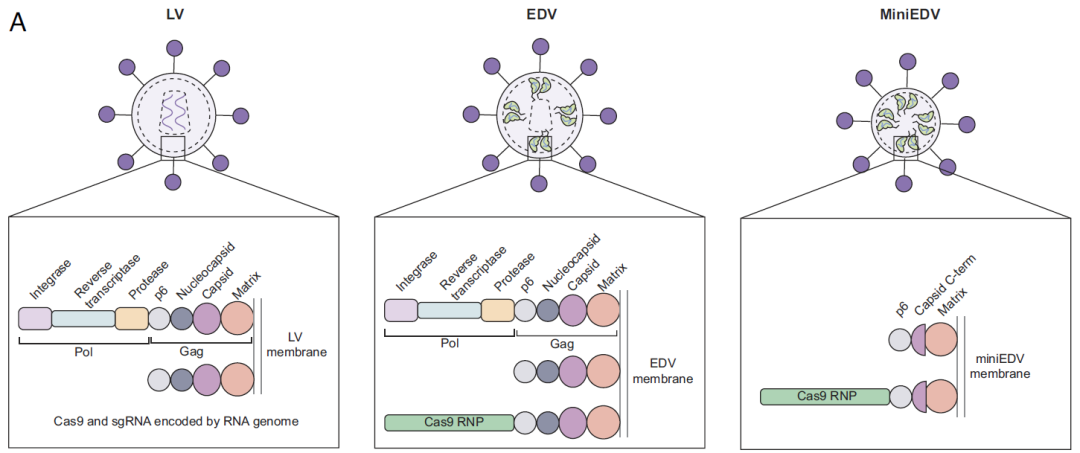
Figure 7: Schematic showing the viral structural proteins in LVs, EDVs, and minimized EDV (miniEDVs)
(Source: Ngo W et al, Proc Natl Acad Sci USA., 2025)
VLP technology is reshaping the landscape of gene editing delivery, with its development exemplifying the innovative pathway of “biomimetic design – engineering optimization – clinical translation”. The ultimate goal of VLPs is to achieve a dynamic balance between precise delivery and biosafety. With ongoing breakthroughs in basic research and the continuous improvement of GMP manufacturing systems, this technology holds promise to bring transformative therapies to fields such as genetic diseases.
Reference:
1 Yin D, Ling S, Wang D, Dai Y, Jiang H, Zhou X, Paludan SR, Hong J, Cai Y. Targeting herpes simplex virus with CRISPR-Cas9 cures herpetic stromal keratitis in mice. Nat Biotechnol. 2021 May;39(5):567-577.
2 Ling S, Yang S, Hu X, Yin D, Dai Y, Qian X, Wang D, Pan X, Hong J, Sun X, Yang H, Paludan SR, Cai Y. Lentiviral delivery of co-packaged Cas9 mRNA and a Vegfa-targeting guide RNA prevents wet age-related macular degeneration in mice. Nat Biomed Eng. 2021 Feb;5(2):144-156.
3 Wei A, Yin D, Zhai Z, Ling S, Le H, Tian L, Xu J, Paludan SR, Cai Y, Hong J. In vivo CRISPR gene editing in patients with herpetic stromal keratitis. Mol Ther. 2023 Nov 1;31(11):3163-3175.
4 Segel M, Lash B, Song J, Ladha A, Liu CC, Jin X, Mekhedov SL, Macrae RK, Koonin EV, Zhang F. Mammalian retrovirus-like protein PEG10 packages its own mRNA and can be pseudotyped for mRNA delivery. Science. 2021 Aug 20;373(6557):882-889.
5 Banskota S, Raguram A, Suh S, Du SW, Davis JR, Choi EH, Wang X, Nielsen SC, Newby GA, Randolph PB, Osborn MJ, Musunuru K, Palczewski K, Liu DR. Engineered virus-like particles for efficient in vivo delivery of therapeutic proteins. Cell. 2022 Jan 20;185(2):250-265.e16.
6 An M, Raguram A, Du SW, Banskota S, Davis JR, Newby GA, Chen PZ, Palczewski K, Liu DR. Engineered virus-like particles for transient delivery of prime editor ribonucleoprotein complexes in vivo. Nat Biotechnol. 2024 Oct;42(10):1526-1537.
7 Ngo W, Peukes J, Baldwin A, Xue ZW, Hwang S, Stickels RR, Lin Z, Satpathy AT, Wells JA, Schekman R, Nogales E, Doudna JA. Mechanism-guided engineering of a minimal biological particle for genome editing. Proc Natl Acad Sci U S A. 2025 Jan 7;122(1):e2413519121.

US: 3675 Market Street, Suite 200, Philadelphia, PA19104 Tel: +1 (215) 205-6963 | +086 027-65023363
E-mail: hui.wang@genevoyager.com / vector@genevoyager.com
China: No128, Guanggu 7th Rd, East Lake High-tech Development Zone, Wuhan, China Tel: 17720522078
E-mail: marketing@genevoyager.com